Complement component 5(C5) is a central hub of the complement cascade, with a molecular weight of approximately 190 kDa, composed of an α-chain (111 kDa) and a β-chain (75 kDa) linked by disulfide bonds. Upon cleavage by C5 convertase, C5 generates two potent effector molecules: C5a, a highly active anaphylatoxin and chemoattractant that drives the recruitment and activation of neutrophils and monocytes; and C5b, which initiates the formation of the membrane attack complex (MAC, C5b-9) leading to target cell lysis. Therefore, blocking C5 simultaneously inhibits terminal complement activation and downstream inflammatory signaling, while preserving upstream C3b-mediated opsonization—making C5 a preferred target in complement-related drug development.
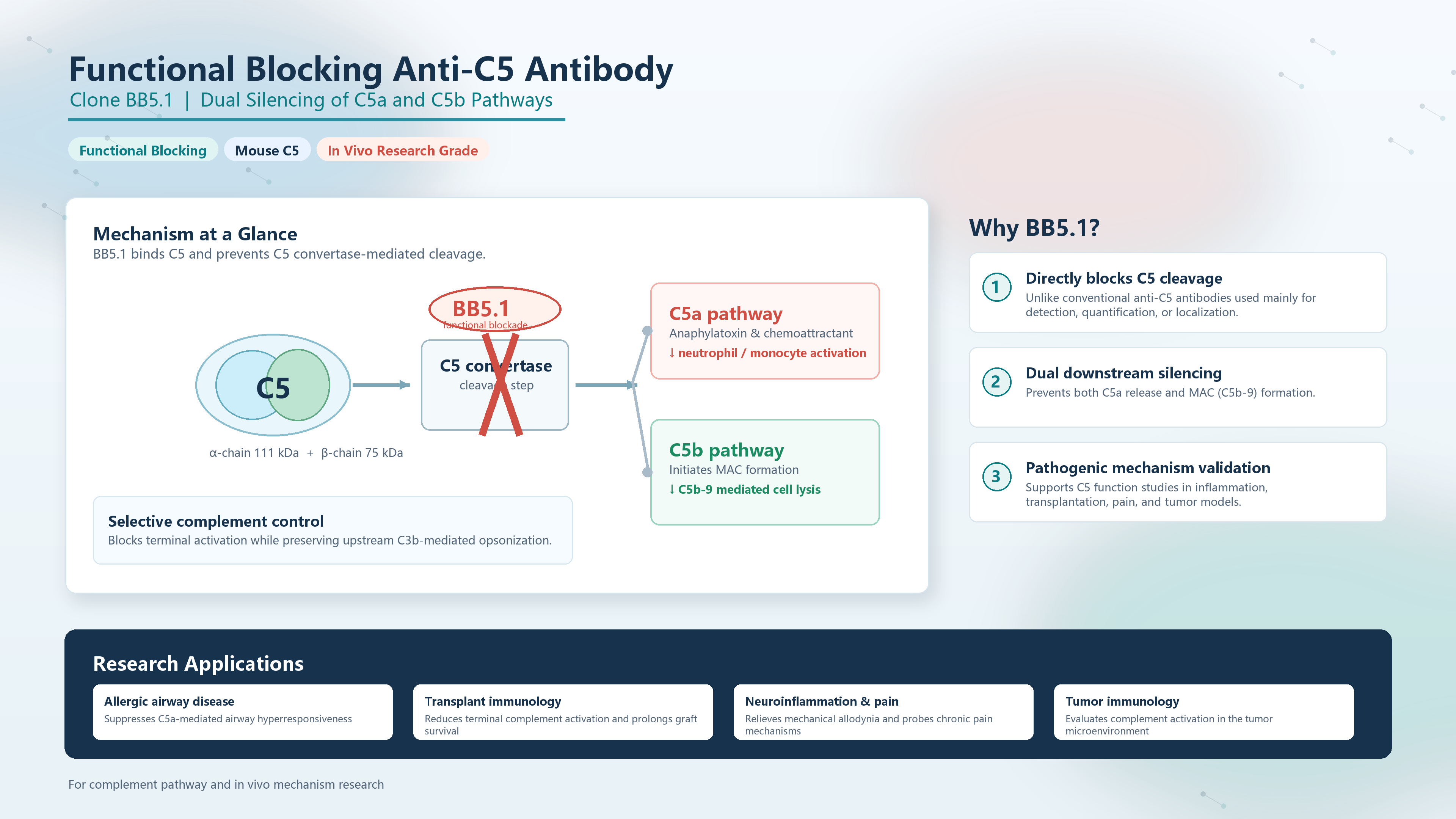
Common anti-C5 antibodies are typically used for detection, quantification, or localization studies, and lack the ability to inhibit C5 cleavage. In contrast, the monoclonal antibody clone BB5.1 is a landmark functional blocking antibody in complement research. BB5.1 binds mouse C5 with high affinity and directly inhibits its cleavage by C5 convertase, thereby preventing both C5a release and MAC formation. This functional capability distinguishes BB5.1 from standard antibodies: while conventional antibodies can “detect” C5, BB5.1 effectively “silences” its biological activity.
The anti-C5 antibody (clone BB5.1) plays a critical role in validating the pathogenic function of C5 in various disease models, including:
Allergic airway disease: BB5.1 effectively suppresses C5a-mediated airway hyperresponsiveness, providing direct evidence for the role of complement in asthma pathogenesis.
Transplant immunology: By blocking terminal complement activation, BB5.1 significantly prolongs graft survival and reduces transplant-associated inflammatory responses.
Neuroinflammation and pain research: Intrathecal administration of BB5.1 alleviates mechanical allodynia, revealing a key role of the complement system in chronic pain regulation.
Tumor immunology: BB5.1 is used to assess the impact of complement activation on tumor growth and the tumor microenvironment.
Guided by the mission of “empowering science,” InvivoCrown focuses on delivering high-purity, cost-effective, function-driven in vivo-grade antibodies for global researchers in immunology and oncology.